Live Longer
Live Stronger
Live Healthier
Experience the Power of Advanced U.S. Stem Cell THERAPY– Right Here in Arkansas.
Joe Rogan – Mel Gibson on How Stem Cell Therapy Saved His Dad’s Hip
CONDITIONS ADDRESSED WITH STEM CELLS AND EXOSOMES

AUTOIMMUNE

NEUROLOGICAL

COSMETIC

CARDIOVASCULAR

KIDNEY AND LIVER

BACK PAIN & DEGENERATION

MUSCLESKELETAL

NERVE & MYELIN SHEATH

ORTHOPEDIC

PULMONARY

REPRODUCTIVE

OTHER

WELLNESS OPTIMIZATION

DENTISTRY
ABOUT MDVISIT STEM CELL LABRATORIES
High-Quality Mesenchymal Stromal Cells (MSCs) and Exosomes from Sweden
GMP-certified mesenchymal stromal cells (MSCs) manufactured in Sweden for pre-clinical and clinical research. MSCs are studied for their anti-inflammatory and immunomodulatory properties. By providing high-quality MSCs at scale, we support ethically approved research collaborations and help advance scientific understanding. Treatment provided in the Bahamas clinic. (Global Services)
High-Quality Mesenchymal Stromal Cells (MSCs) and Exosomes from the United States
FDA-certified laboratory harvesting mesenchymal stromal cells (MSCs) in Florida for pre-clinical and clinical research. MSCs are studied for their anti-inflammatory and immunomodulatory properties. By providing high-quality MSCs at scale, we support ethically approved research collaborations and help advance scientific understanding. Treatment provided in our U.S. clinics and our Mobile services at your home or office. (AR, TN, TX, MO, LA, and MS)
OUR LIST OF REGENERATIVE THERAPY SERVICE

U.S. Laboratory for Stem Cells and Exosomes – Umbilical Cord Derived
In clinic therapy (Joint injections, system IV, microneedling, and nebulizer treatment)
Home and Office Therapy (System IV, Microneedling, and Nebulizer)

Sweden laboratory for Stem Cells and Exosomes – Bone Marrow Derived

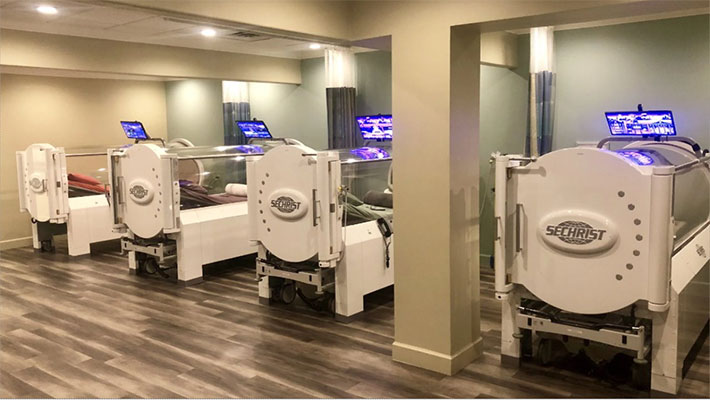
Hyperbaric Oxygen Therapy – Hospital Grade Chambers up to 3.0 ATM

Nuebie Advanced Physical Therapy

Cellcore Advanced Detoxification – pathogen, parasite and toxin elimination

Exercise With Oxygen (EWT) Athlete Advanced Training

STEM CELL THERAPY CONSULTATION
Cellular tissue therapy has many different applications, but understanding your personal situation, and tailoring a therapy accordingly, is important. Schedule a consultation to learn more, and see if cellular tissue therapy is right for you.
- FREE FOR A LIMITED TIME
- Speak with one of our health experts 1-on-1
- Identify the therapy plan that's right for you
- This consult is OVER THE PHONE.
Payment plans for stem cell therapies are available.
MDVISIT CLINICS & THERAPY CENTERS
Our medical directors are in Sweden and Florida. They guide our clinic and therapy center partner on the most current protocols for stem cells, exosomes, and hyperbaric oxygen.
Our clinics and therapy centers then administer the protocols through four (4) different delivery methods. 1) IV therapy, 2) Joint injection, 3) nebulizer, and 4) micro-needling.
Mobile Stem Cell Therapy at Home or Office


What our customers say


What has stood out most to me is not only the integrity of their work, but also their remarkable flexibility and professionalism. Paul has gone above and beyond to accommodate the unique scheduling and therapeutic needs involved in this case—something that is both rare and deeply appreciated.
Collaborating with a like-minded practitioner who understands the importance of integrative, evidence-aligned care has made this process both seamless and rewarding. I look forward to continuing to work with this clinic and highly recommend them to other professionals seeking advanced, compassionate solutions for their clients’ cellular and regenerative health.





Roughly a month after she got stem cell therapy, she was having a really bad day. She grabbed the side of her head and said her brain was freezing! Surprisingly, that was the moment of her turnaround. Since that night, she had been improving like a wild woman!
She can now clearly explain what she needs and, she understands everything going on around her.
Now, she tells me what she wants to eat, watch on TV, and where she wants us to go. Best of all, she now tells me daily that she loves me! She is also now very active and lost the 45 pounds she had gained.
It is by far, the best money I have ever spent in my life!



Recently, I was referred to MDVISIT Stem Cells by a friend. They thoroughly assessed all my symptoms from a combination of conventional medicine and holistic medicine. I was happy to know their rendering providers are Arkansas-licensed medical doctors. It is typically difficult to find an MD who thinks with a holistic mindset.
Now, I am one month out from stem cell and exosome therapy. My pain has decreased 50%. I do understand it takes 3 months to 6 months to feel the full set of improvements because the body tissue needs to grow and regenerate.
I also decided to do their hyperbolic oxygen therapy and their advanced Neubie physical therapy.
Begin your journey to a healthy and independent life.
Here’s how to do it.
1. Apply Today
You can apply for therapy by calling and speaking to a case manager. Upon review and approval by our medical directors, he will craft your personalized care plan for you.
2. Receive Treatment
3. Follow Up
After therapy, your case manager will follow up with you regularly to monitor your progress, answer questions, and provide support on your health journey.
Frequently asked questions
Regenerative medicine involves therapies that repair, restore, or regenerate damaged tissues, offering a major leap beyond traditional treatments that merely mask symptoms. These therapies fall into two categories: those using a patient’s own biologics (like bone marrow or adipose tissue) and those from external sources (such as PRP therapy, amniotic fluid/membrane, or umbilical cord blood/tissue). Misinformation about external-source biologics is common, often spread by industry competitors using skewed data to mislead potential patients.
Amniotic and umbilical cord tissues are sourced from healthy, consenting donors under 35 during planned C-sections, with the process tightly regulated by the FDA to ensure safety through rigorous acquisition, testing, processing, and storage standards.
During a routine C-section, products of conception—such as amniotic fluid, placenta, and umbilical cord—are typically discarded. Instead, with the mother’s consent and the baby unharmed, these materials are donated, placed in a sterile container, and promptly delivered to a nearby FDA-registered, certified lab.
No, biologic materials do not come from “aborted fetuses.” During collection, babies are unharmed, and the donated materials, typically discarded, are used instead.
Additionally, embryonic stem cells are no longer legally used in the US. There’s no fetal tissue or cloning involved, and all donors are thoroughly screened and provide consent per FDA regulations.
This ensures there are no ethical concerns with using these products of conception.
Referred to as “products of conception,” regenerative materials from amniotic and umbilical tissues include:
- Amniotic Fluid
- Umbilical Cord Tissue
- Umbilical Cord Blood
- Wharton’s Jelly
While their precise role in fetal development is beyond this guide’s scope, these materials support fetal growth and translate to patient benefits in regenerative procedures, such as preventing infection and promoting tissue growth (e.g., collagen, tendon, lung, kidney, heart).
Research highlights several benefits of these materials, including:
- High stem cell counts
- Abundant growth factors
- Numerous cytokines
- Other components like exosomes, microsomes, secretomes, and mRNA
A fitting analogy is that these materials form a regenerative “orchestra,” delivering a comprehensive array of components for patient treatment.
Note: Some competitor marketing may claim “products of conception” lack live cells. This can be true if biologics are heavily radiated or over-preserved during processing, but not if processed with minimal radiation and preservatives. This is why MDVISIT CLINIC carefully selects labs to ensure safe, high-quality products for optimal patient outcomes.
The range of conditions that may benefit from amniotic and umbilical procedures is broad and expanding. Under FDA regulations, these biologics can be used for conditions where physicians consider them safe and clinically appropriate. Our disclaimer remains: None of the therapies mentioned have been evaluated by the FDA. As with all medical procedures, MDVISIT CLINIC does not guarantee specific outcomes, and no protocol or biologic indication has been evaluated or approved by the FDA.
- Sports Injuries
- Neurologic Conditions:
- Soft Tissue Conditions: Tendonitis, Bursitis, Ligament Injury (e.g. Achilles, Knee, Rotator Cuff)
- Arthritis: All Types
- Autism
- Migraines and Cluster Headaches
- Post Herpetic Neuralgia
- Diabetes
- Pelvic Pain
- Phantom Limb Pain
- Lyme Disease
- Fibromyalgia
- Heart/Kidney/Lung Failure (e.g. Cardiomyopathy, COPD)
- Back Pain
- Cerebral Palsy
- Trigeminal Neuralgia
- SI Joint Pain
- Erectile Dysfunction
- RSD
- Tennis/Golfer’s Elbow
- Neuropathy
- Plantar Fasciitis
PRP, or platelet-rich plasma, is derived from a patient’s blood draw, placed in a kit, and spun in a centrifuge for 10-15 minutes. This separates the blood into layers, with the middle “buffy coat” layer containing concentrated platelets, white blood cells, and 8-12 growth factors. PRP has minimal, if any, stem cells, making it a valuable regenerative biologic but not a stem cell therapy.
In contrast, amniotic and umbilical cord materials contain over 80 growth factors, along with abundant cytokines, mRNA, exosomes, secretomes, and other elements, including stem cells. The concentration of these components varies by lab, depending on factors like preservatives or radiation used in processing.
No Harvest Required
Bone marrow stem cell procedures involve aspiration from the patient’s iliac crest (pelvis), which carries a 29% risk of chronic pain and potential complications like nerve or vessel injury, bowel perforation, or fracture. Additionally, stem cell quantity and quality in bone marrow decline sharply with age—1 in 10,000 cells at birth versus 1 in 2 million by age 70. Amplifying cell counts through culturing is illegal in the US, and concentrating bone marrow doesn’t solve the low cell count issue.
Adipose-derived stem cell procedures require mini-liposuction from the abdomen or buttock, but many patients lack sufficient fat tissue. While adipose tissue is rich in stem cells, over 80% die within 48 hours after processing and transfer, limiting their effectiveness.
Safety
Bone marrow aspirations have a high complication rate, with 29% of patients experiencing chronic pain—ironic when the goal is pain relief. Other risks include infection, bleeding, nerve/vessel damage, bowel perforation, and pelvic fracture. Mini-liposuction for adipose procedures has fewer complications, but the rapid stem cell die-off undermines its value.
Amniotic and umbilical materials bypass these issues entirely by eliminating the need for invasive harvesting. Amniotic fluid lacks sufficient HLA (MHC) factors to trigger rejection, making it immunologically privileged and safe from Graft-versus-Host reactions. Umbilical cord tissue or blood could potentially cause rejection if not processed correctly, but removing red blood cells eliminates HLA factors, preventing adverse reactions.
Consistency
Amniotic and umbilical materials offer high consistency when processed at FDA-certified labs, delivering robust and reliable cell counts. Unlike bone marrow and adipose, where cell numbers and quality drop significantly with age, these materials maintain their potency. A common myth claims processed amniotic fluid lacks live cells. However, the FDA doesn’t mandate radiation, and with minimal preservatives, cells remain viable. Cryopreservation also preserves cell vitality (as seen in egg donor programs), ensuring labs using careful methods deliver abundant live cells.
Excellent Outcomes
Countless studies demonstrate the effectiveness of amniotic and umbilical tissues for musculoskeletal conditions—PubMed is a great resource, with key references listed at this guide’s end. With a high benefit profile and minimal risk, these therapies are a medical “home run.”
Dr. Arnold Caplan, a pioneer at Case Western Reserve University, is considered the father of modern stem cell therapy. His research reveals that regenerative materials primarily act as signals, prompting the body to “start working” and repair itself. He suggests redefining MSCs—typically Mesenchymal Stem Cells—as Medicinal Signaling Cells.
This shift “better reflects how these cells target sites of injury or disease, releasing bioactive factors that modulate the immune system and promote regeneration, essentially producing therapeutic drugs on-site.” He adds, “It’s the patient’s own local, tissue-specific stem cells that build new tissue, driven by the bioactive factors from the externally provided MSCs.”
MDVISIT CLINIC is growing clinics across the country offering regenerative procedures. If your nearest center doesn’t provide the specific treatment you need, our concierge service can help arrange travel to a center that does.
Regenerative procedures are not currently covered by insurance, but this isn’t because they’re ineffective. Insurance coverage is complex, even for FDA-approved drugs, and amniotic fluid or umbilical cord tissue can’t be patented by pharmaceutical companies.
New technologies often take 5-10 years to gain acceptance for coverage by commercial insurers and Medicare. A positive step is that some states now cover these procedures under Worker’s Compensation claims, as they help patients return to work.
MDVISIT CLINIC ensures finances don’t block access to regenerative therapies, offering various financing options and payment plans.
Substantial research on products of conception has been published in peer-reviewed journals or presented at national professional society meetings. New studies are added monthly, consistently demonstrating the safety of amniotic and umbilical materials.
These materials have also shown promising results for a range of conditions, including musculoskeletal disorders, neurodegenerative diseases, autism, stroke, organ failure, autoimmune conditions, and challenging cases like Lyme disease.
It’s inappropriate for anyone to set unrealistic expectations about these therapies. Terms like “heal” or “cure” are misleading and erode patient trust. No study shows 100% success, and while patients often experience improvement and relief, a “cure” is not guaranteed. When asked if our affiliated providers offer a “guarantee,” the answer is no—medical procedures are never certain. More accurate terms include facilitate, mitigate, or improve.
The biologic components in these materials collaborate to repair damaged tissue and support the body’s own repair processes. The degree of improvement varies because each person is unique.